Pumping Heat Can Be Seven Times More Efficient Than Creating Heat
Abstract: Heat pumping is the simplest, fastest and most effective way to heat up, or cool down, something. Deliberate heat creation by human beings, fire, is around 1.5 million years old. Heating and cooking used burning, which is quick energy releasing oxidation. Around 100,000 years ago, Neanderthals used fossil fuels (lignite) to make fire.
A completely different tech was invented in 1834: Heat Pumping . Commercially available heat pumps in 2022 can be seven times more efficient energy-wise than the maximum available when creating heat by burning.
It is perplexing that states did not make heat pumping mandatory. It may reflect a secret political agenda to remain dependent upon fossil fuels, as the power of most of the world’s potentates is entangled with them.
Real ecologists should be vociferous about heat pumping. Differently from electric vehicles which are of dubious proximal ecological advantage, heat pumping’s benefits are immediate, considerable, pollution free, and without ecological cost.
Heat pumping should be mandatory: it would considerably mitigate the CO2 crisis.
***
Basic Physics of Heat Pump Without Mathematizing:
A heat pump is a device to transport heat, using a gas as a carrier. Heat pumps heat something by bringing energy to create heat, heat pumps trick nature by displacing heat. By displacing heat, heat pumps heat one place and cool another. An application is creating a cold place (refrigerators), another is to create a warm, or potentially extremely hot place.
Heat pumps are an excellent occasion to ponder the nature and relations between heat, temperature and motion. Basically heat is temperature writ large, and temperature is motion. Thus in a way heat is (disordered) motion and it’s not surprising that this motion can be channeled and moved somewhere else.
Heat pumps can be very advantageous: whereas one would need 600 units of energy to create a given heat, by raising the temperature directly, it can take only 100 units of energy to get the same heat, just by transporting the heat to the same place. Intuitively, it’s a bit like needing much less energy to transport a number of people to a place, rather than creating the same people from scratch.
OK, let’s get a bit more technical:
***
PHYSICS OF HEAT PUMPS:
The definition of a gas is that it is a state which can be compressed (and is not a plasma). Heat pumps use the compressibility of gasses crucially.
Heat needs to be defined. Heat is different from temperature. At the molecular scale, temperature in a gas is defined by the average speed of motion of the molecules making the gas. That speed creates (kinetic) energy. When a molecule hits a wall, it transmits its kinetic energy to the wall: that’s heat.
Basically one can say that heat is agitation.
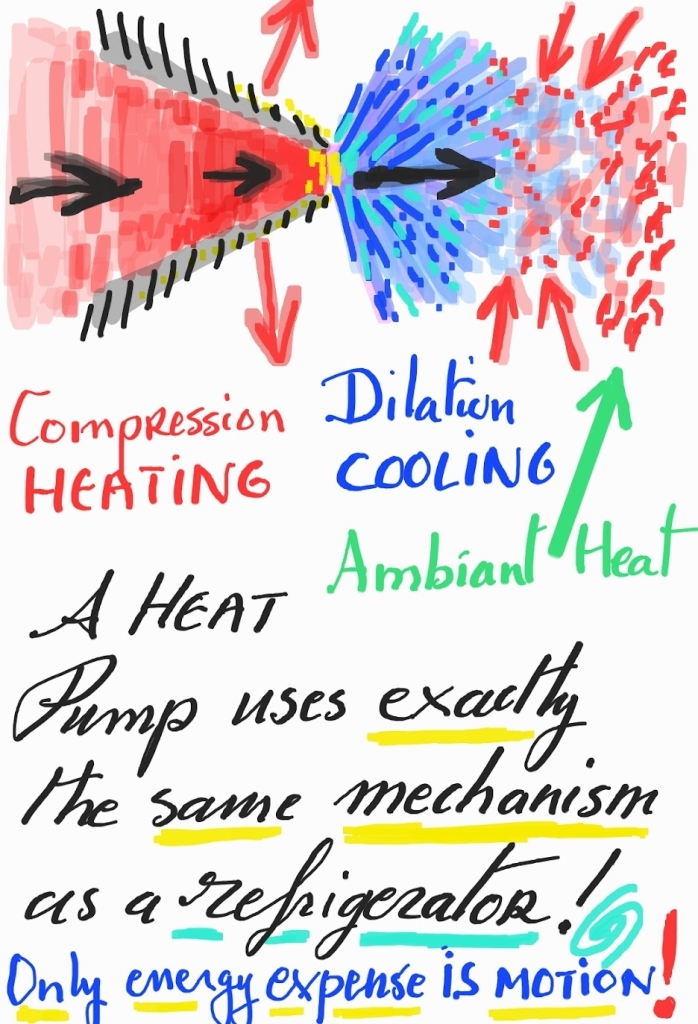
The closer the walls containing a gas, the more frequently the molecules will hit the walls… thus the more heat will be transmitted to said walls. So, as a gas gets compressed, more heat gets transmitted to the walls. Thus, if one pushes a gas through a nozzle, the nozzle will heat up. The effect is boosted because the compression itself raises the temperature of the gas: all the energy put into the compression gets into the gas, and that means it is converted into the form of energy the gas has, and that is kinetic energy… also known as temperature. So compression raises the temperature while transmitting more heat: a double bang.
Let’s suppose the gas can expand after the nozzle: as its momentum gets confined by the nozzle, it will move slowly after it escapes it. In other words, it will be colder than how it started before the compression… And also the compression, although bringing in energy, made it lose even more energy to the walls (if one arranges the walls and their material well). This cooling of the gas enables us to make a refrigerator.
In a heat pump, the gas, exposed to the environment, will then warm up, back to the temperature and pressure it had before the compression. So the gas acquires heat from the environment, and then can be brought to the compressor, which releases it. Now make a closed circuit: the heat taken from the environment will then be released just past the compressor at the nozzle.
Plugging in the numbers and equations, or just experimenting with various gasses, nozzles and compressors, one gets efficiencies in commercially available units, which are seven times the maximum theoretical efficiency limit given by direct heating (given by Carnot law).
Why have heat pumps not be made mandatory? As with High Efficiency Particulate Air (HEPA) filters, which clean the air from all sorts of germs and pollutants, that is a mystery…
Imposing heat pumps is the simplest legislative effort which can be made at this point to mitigate the CO2 crisis.
Patrice Ayme